The adjuvants emerged through an exhaustive screening process that compared multiple molecules head-to-head in different combinations. 
With the adjuvants we selected, we were able to make an RBD-based protein vaccine as effective as a full spike-based mRNA vaccine.” “That’s why people have used the full spike protein, which is harder to produce at scale. “The RBD protein by itself is poorly immunogenic,” says Dowling. It combines the same spike protein fragment - the RBD - with two vaccine adjuvants, molecules that boost the immune response. In the Precision Vaccines Program at Boston Children’s, David Dowling, PhD, Ofer Levy, MD, PhD, and colleagues have created a COVID-19 vaccine formulation that may work especially well in older people. (Image: AdobeStock) Adjuvants for older adults The RBD is the basis for two new protein-based COVID-19 vaccines. The team has filed a patent on their technology and hopes to engage biotech or pharmaceutical companies to take the vaccine into further testing and eventually a clinical trial.Ī crystal structure (left) and space-filling molecular model (right) of the receptor-binding domain (RBD) of the SARS-CoV-2 spike protein bound with a cell’s angiotensin-converting enzyme 2 (ACE2) receptor. It also remained stable and potent for at least seven days at room temperature. In tests, the team successfully freeze-dried the vaccine and then reconstituted it without loss of efficacy. It also elicited strong cellular immunity, stimulating the T helper cells that rally other immune defenses. As reported in the November 2 issue of PNAS, its vaccine elicited strong immune responses against SARS-CoV-2 and its variants in mice, stimulating high amounts of neutralizing antibodies against the RBD protein. To target the antigen-presenting cells, the team engineered the nanobodies to recognize and home to class II major histocompatibility complex (MHC) antigens on the cells’ surface.  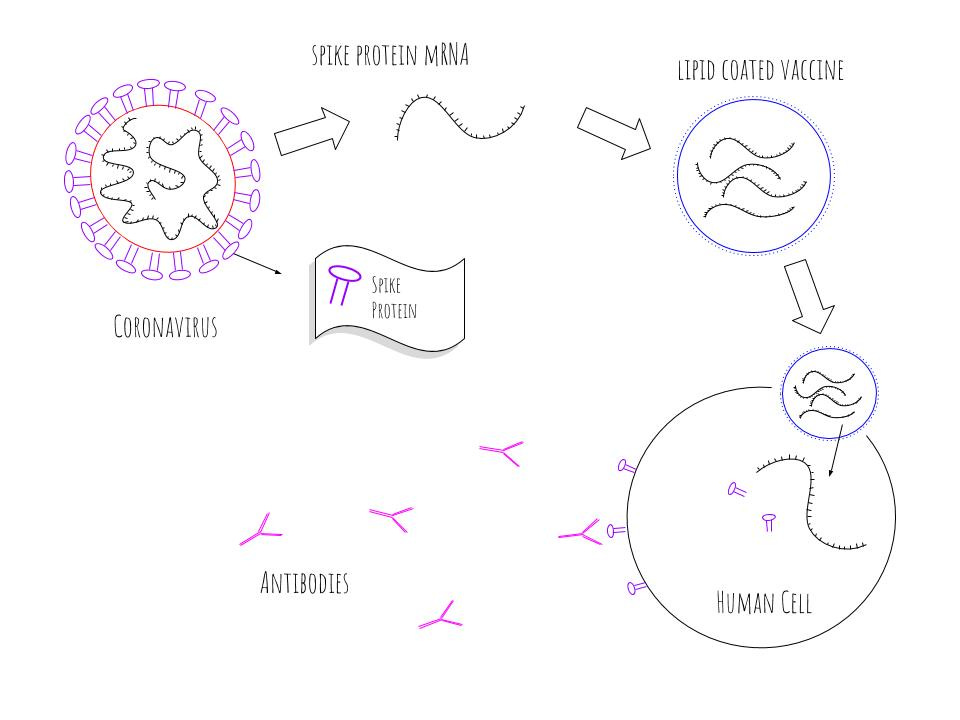
“Taking out the middleman and talking directly to antigen-presenting cells is much more efficient,” he says. This “nanobody,” smaller than human antibodies, steers the RBD protein segment directly to antigen-presenting cells - key immune cells that then “show” RBD to other immune cells, stimulating a broader immune response.Ĭurrent COVID-19 vaccines are presumed to stimulate antigen-presenting cells, but only indirectly, says Ploegh. Alpacas, antibodies, and antigen-presenting cellsĪ team led by Novalia Pishesha, PhD, Thibault Harmand, PhD, and Hidde Ploegh, PhD, in the Program in Cellular and Molecular Medicine, attached the RBD to a special kind of antibody derived from alpacas. But the two vaccines use very different methods to stimulate immune responses. The RBD is the same part that latches onto our cells’ angiotensin-converting enzyme 2 (ACE2) receptors. In contrast, both Boston Children’s vaccines use just a portion of the spike, namely the receptor-binding domain or RBD. This would help get more vaccines to parts of the world like Africa where vaccination rates are currently very low.Įxisting COVID-19 vaccines are based on the full SARS-CoV-2 spike protein. Protein-based vaccines are potentially much cheaper to manufacture at scale than mRNA vaccines and may not require ultra-cold storage. That’s where emerging protein-based COVID-19 vaccines - including two candidates developed at Boston Children’s Hospital - could change the game. But manufacturing costs and the need for ultra-cold refrigeration have limited availability of these vaccines in low-and middle-income countries. (Images: Adobe Stock / Illustration: Fawn Gracey, Boston Children's Hospital)Ĭurrent messenger RNA vaccines appear to offer at least some protection against new SARS-CoV-2 variants, including Omicron, especially for people who have received boosters. A new class of protein-based COVID-19 vaccines are easier to manufacture and may not need ultra-cold storage.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
